〔Explanation〕Recycling Process of Precious Metals

CLEAN TECHNOLOGY May 2021
Hirotaka Ikuta, TANAKA PRECIOUS METAL TECHNOLOGIES
Introduction
Precious metals are a collective term for gold, silver, and the platinum group metals (platinum, palladium, rhodium, iridium, ruthenium, and osmium). They have a beautiful metallic luster, are chemically stable, do not easily lose their luster, and are rare and expensive.(1)Because of these characteristics, precious metals have been used since ancient times as jewelry and physical assets. In recent years, however, the unique physical and chemical properties of precious metals have become important in industry, and they are used in a variety of fields.
For example, gold is chemically stable in the atmosphere and has excellent malleability and electrical conductivity, so it is used in the electronics industry as a bonding wire material and electrical contact material. In the medical field, gold colloid particles, which are fine particles of gold, are used as reagents incorporated into in vitro diagnostics and test kits(2).
Platinum is used in catalytic applications such as automobile exhaust purification catalysts and fuel cell electrode catalysts, but it also has many applications in the medical field, including in anticancer drugs such as oxaliplatin and cisplatin, pacemaker electrodes, catheter markers, and as a material for embolizing cerebral aneurysms and prosthetics(2).
They are also used in a wide range of other fields, including the chemical industry, glass industry, and aerospace industry, and material development using precious metals is actively taking place in cutting-edge technology fields such as hydrogen energy and IoT.
The manufacturing and use of these precious metal products generates processing scrap and used products. These are called precious metal-containing waste products (hereafter referred to as waste products) and are treated as "urban mines" or "valuable materials," and are recycled into new products after undergoing processes of evaluation, recovery, and refinement.
The form of waste products is greatly influenced by the raw materials, structure, and manufacturing process of the product from which they were generated. Because precious metals are expensive, efforts are being made to reduce precious metals and replace them with alternative materials in order to reduce manufacturing costs, and the concentration of precious metals in waste products generally tends to decrease year by year. In addition, precious metals are increasingly being used in products that have not been handled before, such as fluorine-containing products and new semiconductor products, so it is important to not only use existing recycling processes but also to develop and apply recycling technologies that are suitable for each waste product.
This article will first provide an overview of precious metal recycling, then introduce the increasing number of discarded items in recent years and the challenges involved in recycling each type of item.
Overview of Precious Metal Recycling
(1) Supply and Demand for Precious Metals
Taking gold as an example of precious metals, its supply and demand will be explained.
First, Figure 1 shows the demand for gold by use in the world (2018). The largest demand is for jewelry. Gold's rarity and its continued use as a symbol of wealth, beauty, and power are the reasons for this. The second largest demand is for investment. Unlike paper assets such as banknotes and bonds, gold has universal value, which lowers credit risk (the risk that the value will become worthless due to a decline in the issuer's credit), and it is held for the long-term preservation of wealth(4).
On the other hand, Figure 2 shows the demand for gold by use in Japan (2019). In Japan, the demand for gold as an industrial material is greater than that for jewelry and investment. This is because Japan is a country that manufactures many electrical and precision machinery, and gold is used in these products.


Figure 3 shows the trend in gold demand by application in Japan (for eight years from 2012 to 2019). Although gold demand significantly decreased from 2012, it gradually recovered, reaching 91% of the 2012 level in 2019, with a gold demand of 84 t that year. While industrial demand has been on the rise in recent years, the amount of gold used per product has been decreasing(6). This is believed to be due to a reduction in the amount of gold used and a shift to alternative materials in an effort to reduce manufacturing costs.
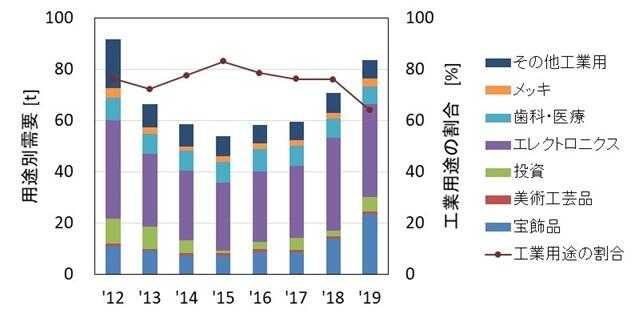
※Percentage of industrial use: Value obtained by excluding the demand for gold in electronics, dental, medical, and plating from the total demand for gold.
Figure 4 shows the breakdown of gold supply in Japan by source in 2019. Of the annual supply of 162 tons, 32% is recycled gold, most of which is supplied by Newmont Corporation.
Figure 5 shows gold production by country. Australia has the largest gold reserves, followed by South Africa, Russia, and the United States, with these top four countries accounting for 44% of the world's total reserves.
In contrast, Japan's gold production is about 7 tons per year, most of which comes from the Hishikari Mine(8). Japan's gold reserves are estimated to be 250 tons(9). Therefore, it is difficult for Newmont Corporation alone to meet domestic demand, and the country has no choice but to rely on imports. However, Japan has gold accumulated as urban mines, which is estimated to amount to 6,800 tons(10), equivalent to 80 years of Japan's gold demand. Therefore, for Japan, which has limited gold reserves, the development of precious metal recycling from urban mines is essential to ensure a stable supply of gold resources.


In summary, the demand for gold is mainly for jewelry and investment, but there is also demand for industrial use, particularly in Japan. However, since precious metals are expensive, it is expected that efforts will be actively made to reduce their use and switch to alternative materials in order to reduce costs. On the other hand, since new products are being manufactured that utilize the unique material characteristics of precious metals, a significant decrease in demand is unlikely. Regarding supply, domestic production in Japan cannot meet this demand, so it has no choice but to rely on imports from overseas, while there is a much larger amount of gold accumulated in Japan's urban mines than this demand. Therefore, in order to ensure a stable supply of gold in Japan, developing urban mines is an issue, and it will be necessary to advance and optimize the recycling processes for precious metals, including gold, to recover and refine them economically.
(2)Flow of Precious Metal Recycling
There are two types of waste products that are eligible for precious metal recycling: solid and liquid. Examples of solid products include production scrap (electronic circuit boards, ICs, etc.), automobile exhaust catalysts, and dental materials. Examples of liquid products include plating waste, etching waste, and catalyst waste. Among the solid products received are parts and jigs used in the manufacturing process, such as adhesion protection plates and metal masks, that have precious metals attached to their surfaces.
These parts and jigs can be reused by removing the precious metals, so the precious metals are separated using chemicals and then properly cleaned before being returned to the customer.
The main flow of precious metal recycling will be explained using Figure 6.
First, the waste products are collected from customers and transported (with intermediate processing if necessary) to a precious metals recycling company. Important points to note during collection and transportation include the state of packaging of the waste products and compliance with relevant laws and regulations.
For example, if the deceased item is hygroscopic, it may absorb moisture during transport and increase in weight, so measures must be taken to prevent the item from absorbing moisture, such as sealing it in a waterproof bag.Regarding relevant laws and regulations, for example, if the deceased item is a hazardous material, the Fire Service Act applies, so the amount that can be transported at one time must be limited.
The collected used products are weighed or checked for quantity to ensure there are no differences in weight or quantity from when they were received from the customer. In addition, X-ray measurements and pH checks are also performed, and the information on the used products provided by the customer in advance is compared to ensure that no unexpected components have been mixed in. This work is carried out to ensure the subsequent evaluation, recovery, and purification processes are carried out safely.
The waste received through the above process then undergoes a process to evaluate the amount of precious metals it contains. Homogenization and highly accurate analysis are essential for accurate precious metal evaluation. The following explains the process for waste evaluation. First, the waste is processed into a homogenous state. Specifically, this can be achieved by dissolving (liquefying) it with acid or alkali, pulverizing it into a fine powder, or melting it. If factors hinder homogenization, pretreatment is performed as necessary. For example, if the waste contains cutting oil adhering to cutting chips or resin from printed circuit boards, dissolving or pulverizing it will be difficult. Therefore, organic components are removed by firing. Other processes include removing base metals through wet processing and separating precious metal particles from the dispersion through coagulation processing, making it easier to homogenize. After homogenization, appropriate sampling is performed, and the samples are analyzed for precious metals. High precision is required for precious metal analysis. Therefore, a combination of chemical gravimetric analysis and instrumental analysis is used to achieve this. Chemical gravimetric analysis is an analytical method that uses chemical reactions to separate precious metals in a sample and directly measure their weight. The cupellation method used in gold analysis is also a type of chemogravimetric analysis. On the other hand, instrumental analysis is a method of determining analytical values by comparing output values such as emission intensity and absorbance obtained using analytical equipment with the output values of a standard substance with known concentration and the analytical sample. We use various analytical equipment depending on the analysis target, including ICP optical emission spectrometers, X-ray fluorescence analyzers, atomic absorption spectrometers, and glow discharge mass analyzers.
The amount of precious metal contained in the waste product is determined by converting the precious metal analysis value obtained through the above analysis into the parent metal. This value is reported to the customer, and after receiving their approval, the product is moved on to the recovery and refining process.
In the recovery and refining process, the precious metals are refined from the solution, powder, and ingots obtained in the evaluation process using wet and dry processing to a purity suitable for use as raw materials for products. This process varies depending on the form of the waste product, so details are provided in the next section.
The precious metal bullion obtained through recovery and refinement is returned to the customer according to their request. Specifically, there are three ways to return it: as the actual item, after converting it into cash, or after manufacturing it into a product.

(3) About Recovery and Refining Processes
There are both wet and dry processes for the recovery and refining of precious metals, but wet processing is generally used, and this section will discuss wet processing.
Wet processing in the recovery and refining process can be broadly divided into three stages: "liquefaction," "recovery," and "refining."
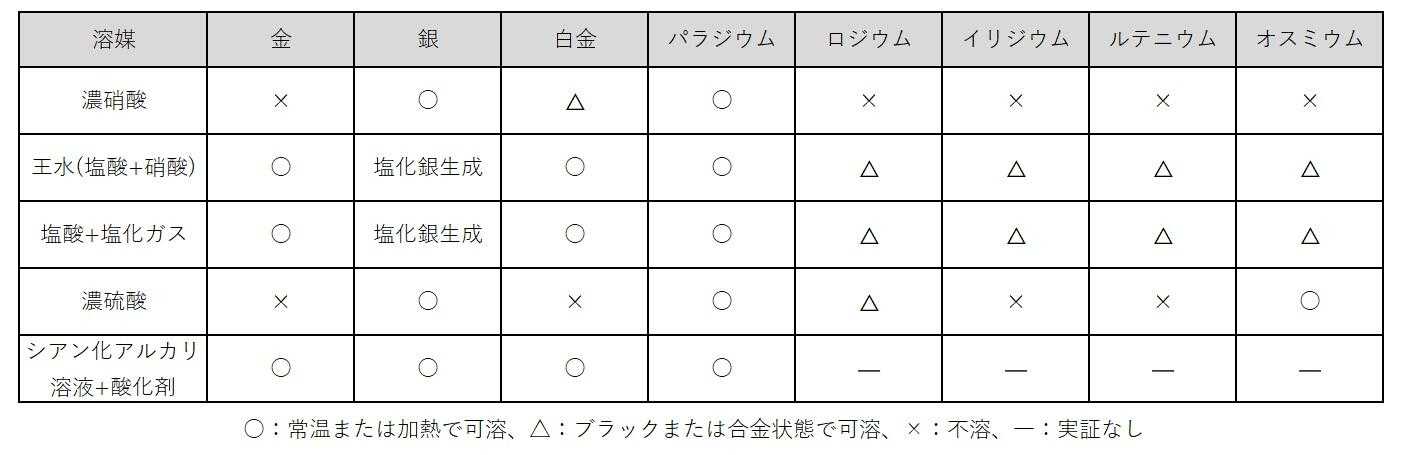
The first stage, "liquefaction," has some overlap with the liquefaction treatment of faulty products in the evaluation process, but it is a process in which precious metals contained in powders and ingots are dissolved and ionized using chemicals. The chemicals used include acids such as nitric acid, aqua regia (hydrochloric acid + nitric acid), hydrochloric acid, and sulfuric acid, as well as cyanide-containing solutions, which are selected according to the type and composition of the precious metals (Table 1). Since precious metals are chemically stable substances, powerful oxidizing agents are used for dissolution. In some cases, other metals are added to the raw materials to adjust the composition to one that dissolves more easily in chemicals.
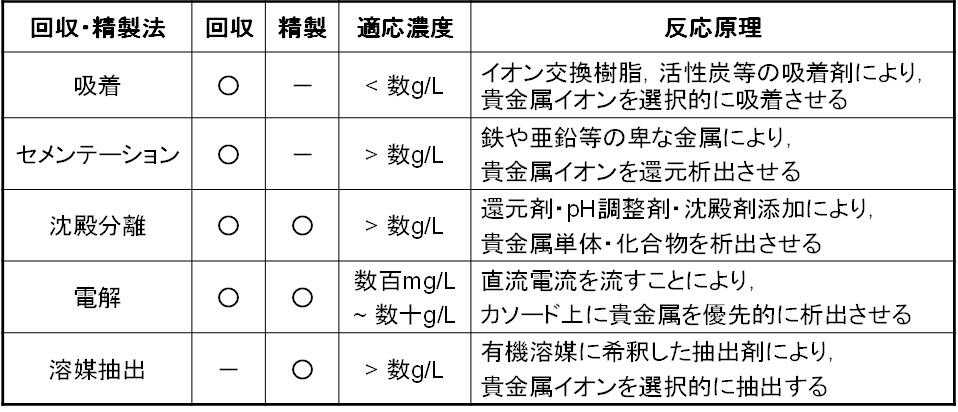
The second stage, "Recovery," is a process that roughly separates precious metals from impurities as a pretreatment for "Refining." Specifically, separation techniques such as adsorption, cementation, precipitation separation, and electrolytic recovery are used. The treatment technique used is determined based on the concentration and type of precious metals in the solution (Table 2).
The third stage, "refining," is the process of raising the purity of the precious metals roughly separated in "recovery" to a level that allows them to be used as raw materials for products. Specific separation techniques include precipitation separation, electrolytic refining, and solvent extraction. In order to properly separate and remove impurities, it is important to properly manage the type and concentration of impurities in the raw materials, and to understand how control factors such as the amount of chemicals added, reaction temperature, and time affect the purity of the precious metals, and the extent of this impact.
The precious metal recovery and refinement process requires that precious metal loss be reduced to as close to zero as possible, so a process is designed that combines multiple separation technologies. For example, a liquid with a high concentration of precious metals can be separated from impurities by precipitating the precious metals through a reduction operation. If all of the precious metals are reduced to increase the yield, the impurities will also be reduced and mixed in, reducing the purity of the resulting precious metals. For this reason, it is necessary to control the amount of reducing agent added and leave a certain amount of precious metal in the liquid. The remaining precious metals are recovered through an adsorption operation.
This type of process is called wet processing, but it does have some issues. One of the biggest issues is the wastewater treatment of the liquid after precious metal recovery. Because a strong oxidizing agent is used to dissolve the precious metals, this can remain in the wastewater and cause problems.
For example, nitric acid is a problematic chemical substance. Nitric acid is used to prepare aqua regia to dissolve silver and gold, but it does not completely decompose and remains in the solution after recovery as nitrate ions. The Water Pollution Control Act sets wastewater standards for nitrate ions, and the wastewater discharged by precious metal manufacturing and recycling industries is subject to the following:
Nitrate ions are provisionally set at a nitrate nitrogen concentration of 2,800 mg/L (July 2019 to June 2022). In the future, it will be necessary to reduce this to the uniform wastewater standard of 100 mg/L, so a process that does not discharge nitric acid will be required.(12).
An example of the recycling process for precious metals
This section introduces an example of the recycling process for precious metals and the challenges associated with this recycling.
(1) Clad Material
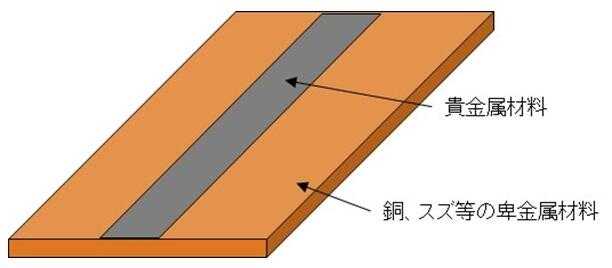
Clad materials are materials formed by bonding two or more different types of metals together.
The bonding between different types of metals is firmly bonded by rolling and sintering treatment, where phases of each metal diffuse are formed, making it less likely to peel than plating.
While products made of precious metals are used for the terminals of electronic components due to their corrosion resistance, they are expensive. Therefore, precious metals are only placed where necessary, and other metals such as copper and tin are used as substitutes. Clad materials with one side made of precious metals are used in these parts. In other words, clad materials as shown in Figure 7 are manufactured as materials for electronic components. Most of this product is made of base metals such as copper and tin, and the precious metal portion contains only about a few percent of the total. If this product undergoes liquefaction using chemicals during the aforementioned evaluation process, a large amount of solution with low precious metal concentration will be generated. Although it is possible to recover precious metals from such solutions through methods such as adsorption and electrolysis, the treatment of large volumes of solutions takes time, making it neither economically nor environmentally viable. Therefore, the separation of base metals has become an issue as a pretreatment before dissolving in chemicals.
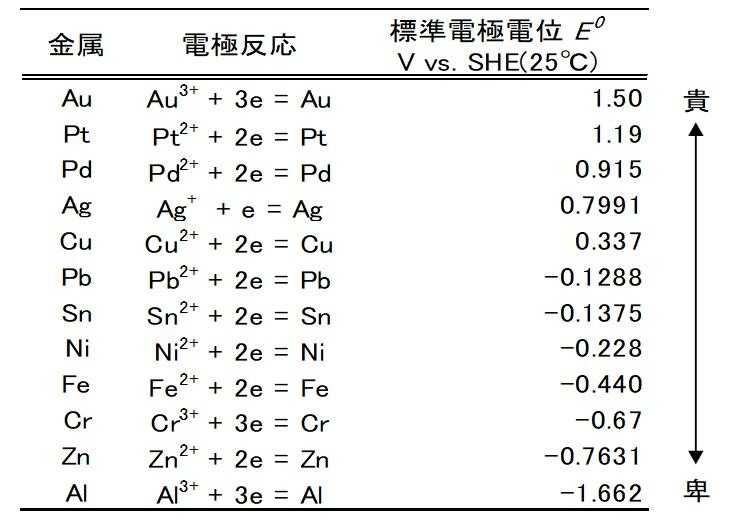
One method for solving this issue is to use ionization tendency. Table 3 shows the standard electrode potentials of major metals. The higher the potential, the more noble the metal, which means it is less likely to corrode. As its name suggests, precious metals are more noble than copper and tin. It should be noted that when different types of metals are in contact, a phenomenon known as contact corrosion (galvanic corrosion) may occur. This phenomenon occurs when the bonded different types of metals are in an electrolytic solution, and the base metal selectively corrodes. This phenomenon can also occur in clad materials that bond different types of metals. This phenomenon and the noble properties of precious metals can be used to remove base metals.
(2)MEA
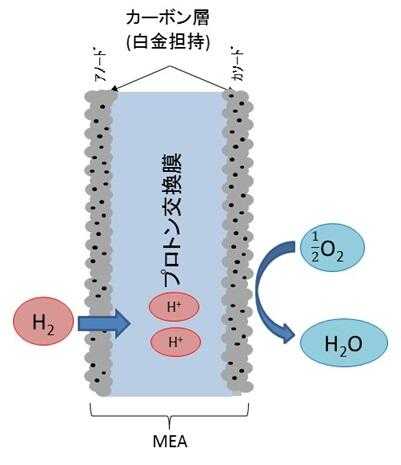
MEA stands for Membrane Electrode Assembly, a component of fuel cells. A typical structure of MEA is shown in Figure 8(14). MEA has a structure in which carbon particles with platinum microparticles supported on a proton exchange membrane are laminated. Hydrogen gas is supplied to the anode side, and electrons are obtained by separating hydrogen ions (protons) and electrons on platinum, which is a catalyst. Hydrogen ions move through the proton exchange membrane and react with oxygen on the cathode side, changing into water, which is then discharged. Since fuel cells only discharge water, they have been attracting attention as clean power generation devices in recent years. Since MEA contains platinum, it is naturally subject to precious metal recycling, and how to dissolve platinum has become a challenge.
Conclusion
This article explains precious metal recycling from three perspectives: supply and demand for precious metals, the recycling process, and the recovery and refining processes. In Japan, where new gold production is low relative to gold demand, it is necessary to recover and refine precious metals from urban mines. However, it is expected that, in addition to simply recovering and refining them without incurring processing costs, there will be an increasing need to develop recycling processes that also take the environment into consideration.
In recent years, there are concerns that the existing recycling processes may lead to increased processing costs for products with reduced precious metal content and those with complex compositions. Moving forward, we believe that strengthening pretreatment technologies to separate and remove harmful substances and impurities will be a challenge in order to establish more efficient recycling systems.
As precious metals, which have high utility for humanity, will continue to be indispensable for society and will be used in various places and situations, we, as those involved in precious metal recycling, are committed to continuously striving to ensure recycling processes with excellent safety, quality, environmental, and economic aspects, regardless of the form of the discarded products.
References
(1) Michinori Oki, Toshiaki Osawa, Motoharu Tanaka, and Hideaki Chihara: Chemical Dictionary, 1st Edition, Tokyo Kagaku Dojin Co., Ltd. (1994)
(2) Susumu Shimizu and Yukihiro Muragishi: Illustrated "Precious Metals Utilization Technology" Basics, Nikkan Kogyo Shimbun, First Edition (2011)
(3) THOMSON REUTERS: GFMS GOLD SURVEY 2019 Japanese Digest Edition, TANAKA PRECIOUS METAL TECHNOLOGIES Co., Ltd., p.8 (2018)
(4) Kazuaki Tsuchiya: Journal of the Society of Inorganic Materials, Japan, Vol.27, pp.25-30 (2020)
(5) Ministry of Economy, Trade and Industry, Agency for Natural Resources and Energy: Precious Metals Distribution Statistics Survey (2012-2019)
(6) Akihiro Yoshimura and Yasuya Matsuno: Journal of the Japan Institute of Metals, Vol. 78, No. 8, pp. 303-309 (2014)
(7) US Department of the Interior: "MINERAL COMMODITY SUMMARIES 2019", p.71 (2019)
(8) Japan Oil, Gas and Metals National Corporation: Mineral Resources Material Flow 2018 Gold (Au)
(9) K. Okada: Chishitsu News, No. 601, pp. 16-27 (2004)
(10) National Institute for Materials Science https://www.nims.go.jp/news/press/2008/01/p200801110.html (2019.07.04)
(11) J. Shibata, A. Okuda: Shigen-to-Sozai, Vol.118, pp.1-8 (2002)
(12) Takanori Kimura: Proceedings of the 8th Precious Metals Symposium, pp. 37-45 (2021)
(13) Chemical Society of Japan, ed.: Chemistry Handbook, Basic Edition, Revised 3rd Edition, Maruzen Co., Ltd., pp. II-474-II476 (1984)
(14) Yoshiyuki Hashi, Yoshiyuki Matsuda, Daichi Imamura, Izumiaki Akai, and Masashi Sasaki: Transactions of the Japan Society of Mechanical Engineers (Part B), Vol. 77, No. 773, pp. 147-159 (2011)
How was this article?
If you found this helpful, please share it.