Diagnostics
What is "Diagnostics"?
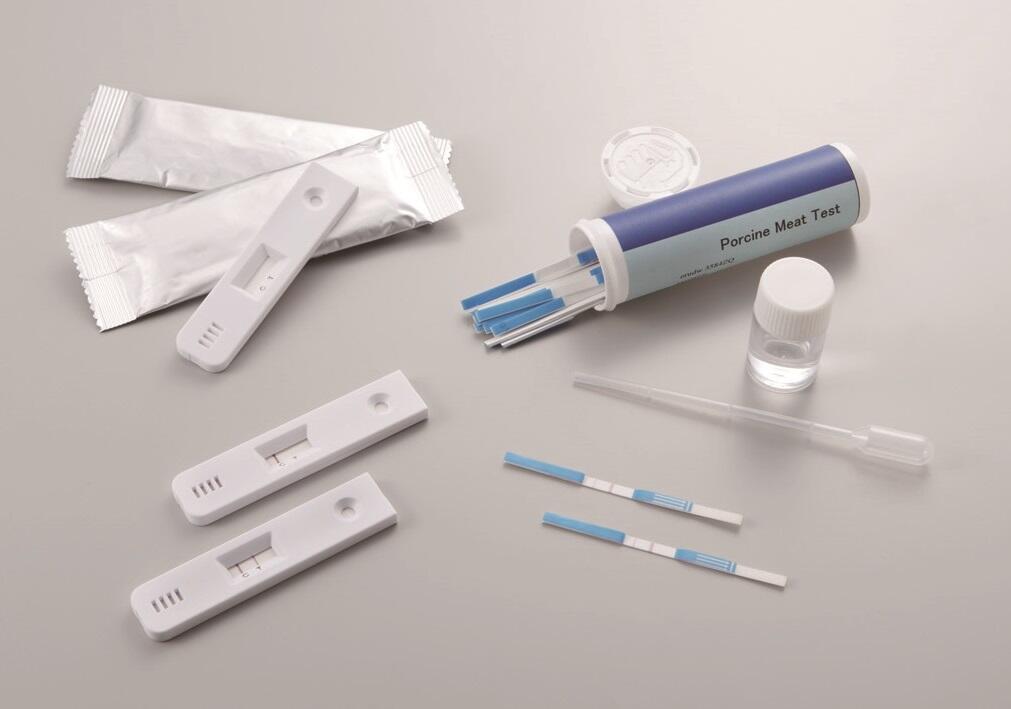
This immunochromatographic in vitro diagnostic reagent utilizes technology to control the particle size of gold colloids at the nano level, enabling rapid and highly sensitive detection of infectious diseases such as influenza and the new coronavirus. We are able to manufacture high-performance reagents by combining high-precision protein immobilization technology, suppression of nonspecific adsorption, and technology to enhance antigen-antibody reactions.
We manufacture in-vitro diagnostic and other test kits using precious metal colloids on a consignment basis.
In addition to the gold colloid manufacturing technologies it has built up over a number of years, TANAKA has at its disposal every possible technology relating to the highest performance immunochromatographic diagnostic test kits. These technologies include those which allow proteins to bind to gold colloids, prevent non-specific absorption of proteins by gold colloids, and promote antigen-antibody reactions.
TANAKA not only supplies raw materials for in-vitro diagnostic test kits, but also manufactures them on an original equipment manufacturer (OEM) basis under a reliable production system managed in accordance with the ISO 13485 system.
We provide products under a reliable production system managed in accordance with ISO13485, from supplying raw materials for in vitro diagnostics to OEM manufacturing.
Consignment details
- Conjugated Colloidal Gold Solution
- Development and manufacturing of test strips for inspection (immunochromatography method※)
- OEM manufacture


Registration and certification
- Registered for the manufacturing of in-vitro diagnostics
- ISO 13485 certified

*Detection principle using immunochromatography method
If antigens (such as virus antigens) are present in a specimen, gold colloids bind to the gold colloid-labeled antibodies to form composite bodies. The composite bodies then pass through a membrane as a result of the capillary action and bind to the capture antibodies that are immobilized in the detection area. These then appear as a red band.
This principle is used to determine whether antigens are present or not.
![[Diagram of Test Strip (Flow of Sample)] From the left: Sample, Colloidal Gold, Colloidal Gold-labeled Antibody, Capture Antibody](https://data.wovn.io/ImageValue/production/688b3c307ffdea2de8055986/en/75dc580b8716f90ec127674112a4399c/wp_content_uploads_sites_images_ex_en_products_images_f03_img_block02_01.jpg)





Related Information
●Disclaimer
Never use this test kit as the only guide to manage your condition or illness. Consult your healthcare provider if your symptoms persist or become more severe, or if you are concerned at any time.
First, please make an Inquiry about the product.
For any questions regarding product specifications, pricing, delivery times, etc., please feel free to Inquiry here.


