Ammonia-Related Catalysts

What are "Ammonia-related Catalysts"?
These precious metal catalysts oxidize and remove low-concentration ammonia emitted from ammonia cracking and other applications, which are attracting attention as a clean energy source.As ammonia is increasingly used as a hydrogen carrier and fuel, this catalyst lineup supports next-generation energy technologies by contributing to efficient reaction promotion and reduced emissions of harmful substances.
TANAKA's Ammonia (NH3) - Related Catalysts
Ammonia (NH3), which is expected to serve as a hydrogen carrier and a zero-emission fuel that, like hydrogen, does not emit CO2 during combustion. We offer ammonia reforming catalysts for decomposing ammonia to generate hydrogen, as well as catalysts for suppressing and decomposing nitrogen oxides (NOx) and nitrous oxide (N2O) in ammonia-containing exhaust gas.
Overview of Ammonia-related Catalysts
Features
- Provides various catalysts based on ruthenium and platinum
- High durability from use of precious metals
- High catalytic activity can be maintained even with low precious metal content, enabling the supply of catalysts at low cost
Main product lineup
| Reaction | Product name | Precious metals | Shape | Particle size (sieve sorting) |
|---|---|---|---|---|
| Ammonia Reforming Catalyst | TRC10-GA Series | Ru | Pellet | Φ approx. 2mm |
| Ammonia Purification Catalyst | APC1-30 Series | Pt | Honeycomb | – |
| Nitrogen Oxides Removal Catalyst | APC1-30 Series | Pt | Honeycomb | – |
- We request for the signing of a Non-Disclosure Agreement (NDA) when providing samples and information such as precious metal content.
- We also accept consultations regarding the prototyping and made-to-order production of customized catalysts to meet customer requests.
Ammonia Reforming Catalyst
These catalyts help to generate H2 fuel from ammonia (NH3)
Reaction: 2NH3→ N2+ 3H2


Characteristics of Ammonia Reforming Reaction
TRC10-GA〔Ru〕
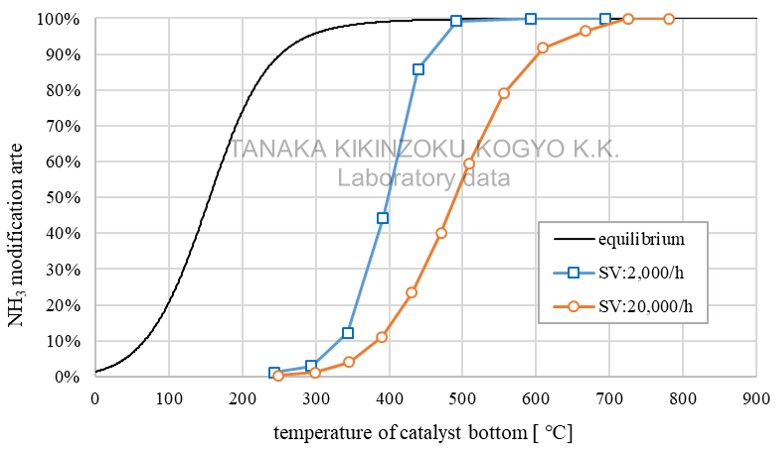
| Item | Value (Unit) |
|---|---|
| Gas Type | NH3 |
| SV (wet) | 2,000 /h 20,000 /h |
Possible to generate hydrogen by reforming ammonia at low temperatures
- Data for reference from laboratory experiments
- This is approximate data as results are affected by factors such as catalyst evaluation conditions, manufacturing conditions and usage environment.
Ammonia Purification Catalyst
Suppresses the generation of nitrogen oxides (NOx and N2O) during ammonia (NH3) decomposition
Reaction formula: 4NH3+ 3O2→ 2N2+ 6H2O

Ammonia purification reaction characteristics
APC1-30〔Pt〕
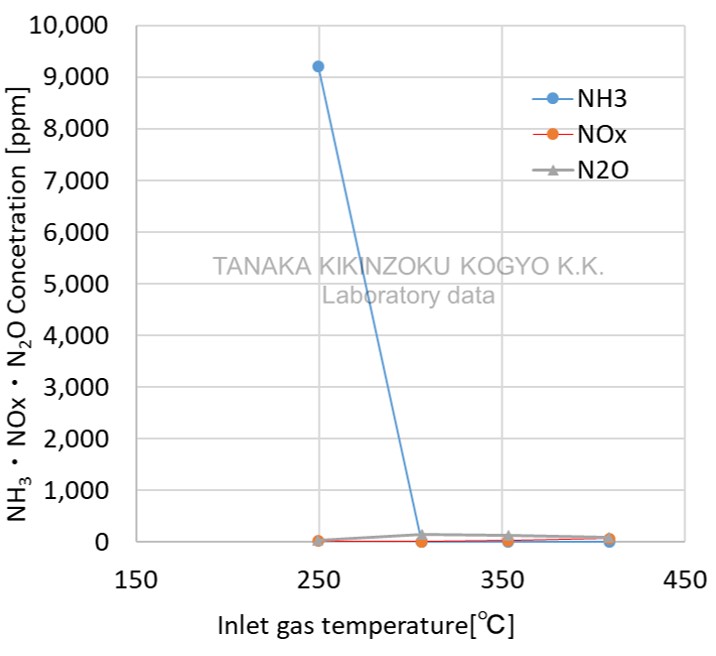
| Item | Value (Unit) |
|---|---|
| NH3 | 1% |
| H2O | 5% |
| エア | Balance |
| SV (wet) | 60,000 /h |
Ammonia decomposition with low NOx and N2O
- Data for reference from laboratory experiments
- This is approximate data as results are affected by factors such as catalyst evaluation conditions, manufacturing conditions and usage environment.
Nitrogen Oxides Removal Catalyst
Ammonia is used to achieve NOx decomposition at low temperatures
Reaction formula: 4NO + 4NH3+ O2→ 4N2+ 6H2O

NOx removal reaction characteristics
APC1-30〔Pt〕
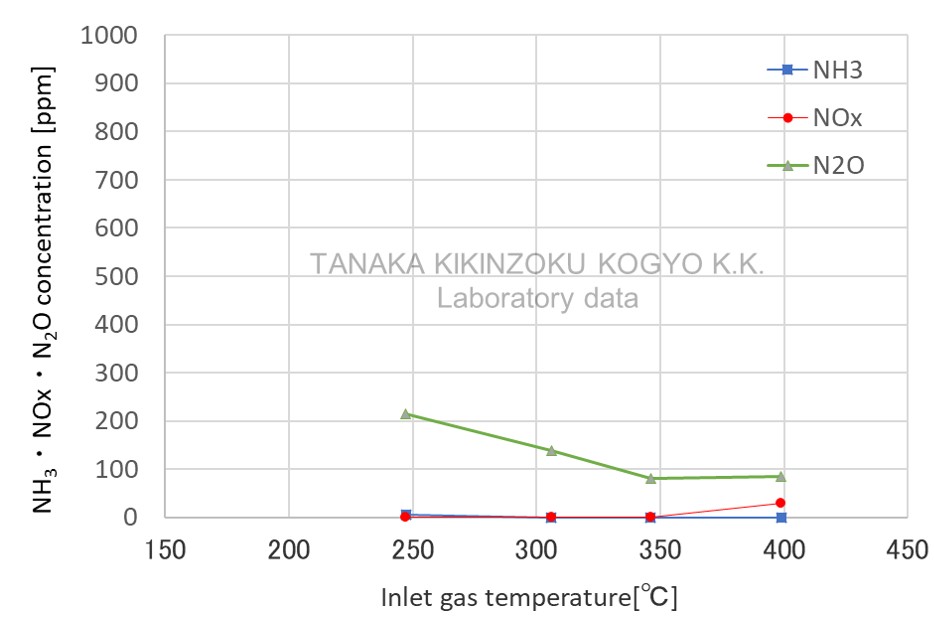
| Item | Value (Unit) |
|---|---|
| NH3 | 2,000 ppm |
| H2O | 5% |
| NO | 1,000ppm |
| エア | Balance |
| SV (wet) | 30,000 /h |
NOx removal at low temperatures is possible
- Data for reference from laboratory experiments
- This is approximate data as results are affected by factors such as catalyst evaluation conditions, manufacturing conditions and usage environment.
Related Information
Please Inquiry
Please feel free Inquiry with any inquiries regarding products, media, competitive funding, etc.





